Redox
|
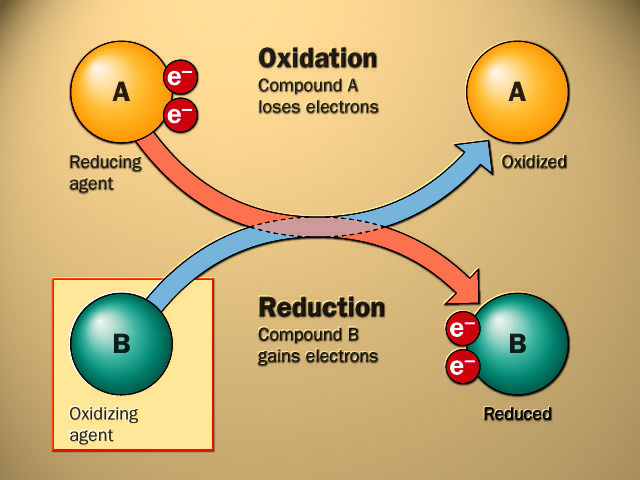
Redox reactions are reactions where reduction and/or oxidation occurs during the chemical reaction
Oxidation reaction is when a species:
Reduction reaction is when a species:
When dealing with electrons an easy way to remember which way round it goes is OILRIG Oxidation Is Loss Reduction Is Gain In these reactions the element that undergoes oxidation is the reducing agent and visa versa |
|
Half Equations
Redox reactions can be transfer of electrons, so can also be called electron transfer reactions.
By separating the reaction in half you can see which species is reduced and which one oxidised.
By separating the reaction in half you can see which species is reduced and which one oxidised.
Sometimes there are more than two ions in the equation:
Oxidation States
Shows which element has been oxidised and reduced in an ionic compound
As an atom or molecule the atom is
Mg2+ has an oxidation state of +2 therefore it has been oxidised
Cl- has an oxidation state of -1 therefore it has been reduced
All the oxidation states add up to 0 in a compound
Ones to remember
Hydrogen is +1 except as a hydride ion where it is -1
Oxygen is -2 except in hydrogen peroxide where it is -1 or F2O where is is +2
Group 7 is generally -1 (Fluorine is always -1)
Group 1 is mostly +1
Group 2 is mostly +2
Aluminium is +3
These may differ because in a compound the first one to be given an oxidation state is the most electronegative - refer to Bonding
For example, CO2 :
Oxygen is most electronegative so:
O = -2
Using algebra, C can be found
C + (2 x -2) = 0
C = +4
As an atom or molecule the atom is
Mg2+ has an oxidation state of +2 therefore it has been oxidised
Cl- has an oxidation state of -1 therefore it has been reduced
All the oxidation states add up to 0 in a compound
Ones to remember
Hydrogen is +1 except as a hydride ion where it is -1
Oxygen is -2 except in hydrogen peroxide where it is -1 or F2O where is is +2
Group 7 is generally -1 (Fluorine is always -1)
Group 1 is mostly +1
Group 2 is mostly +2
Aluminium is +3
These may differ because in a compound the first one to be given an oxidation state is the most electronegative - refer to Bonding
For example, CO2 :
Oxygen is most electronegative so:
O = -2
Using algebra, C can be found
C + (2 x -2) = 0
C = +4
Balancing Redox Reactions
You may be asked to balance an equation. To do this you need to find out which elements are reduced or oxidised and then balance the electron exchange.
For an equation to balance:
E.g.
For an equation to balance:
- Number of atoms of each element on either side must remain the same
- Total charge on either side must equal
E.g.
It gets more complicated in Aqueous Solutions
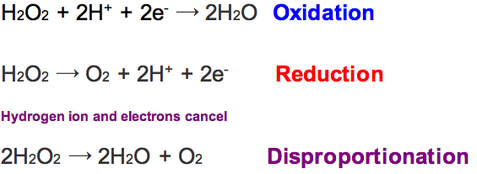
The electrons cancel out
Disproportionation
When a substance undergoes oxidation and reduction simultaneously
Examples are H2O2 and ClO-
Examples are H2O2 and ClO-