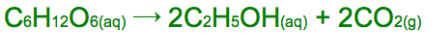Alcohols
Nomenclature
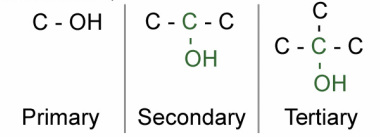
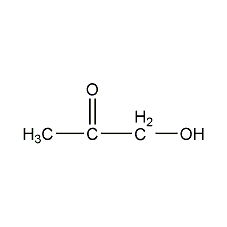
Alcohol
CnH2n+1OH e.g. methanol or propan-2-ol or prefix in alkenes hydroxy
The OH allows for hydrogen bonding - means high melting point
CnH2n+1OH e.g. methanol or propan-2-ol or prefix in alkenes hydroxy
The OH allows for hydrogen bonding - means high melting point
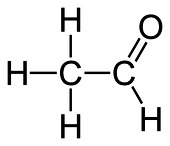
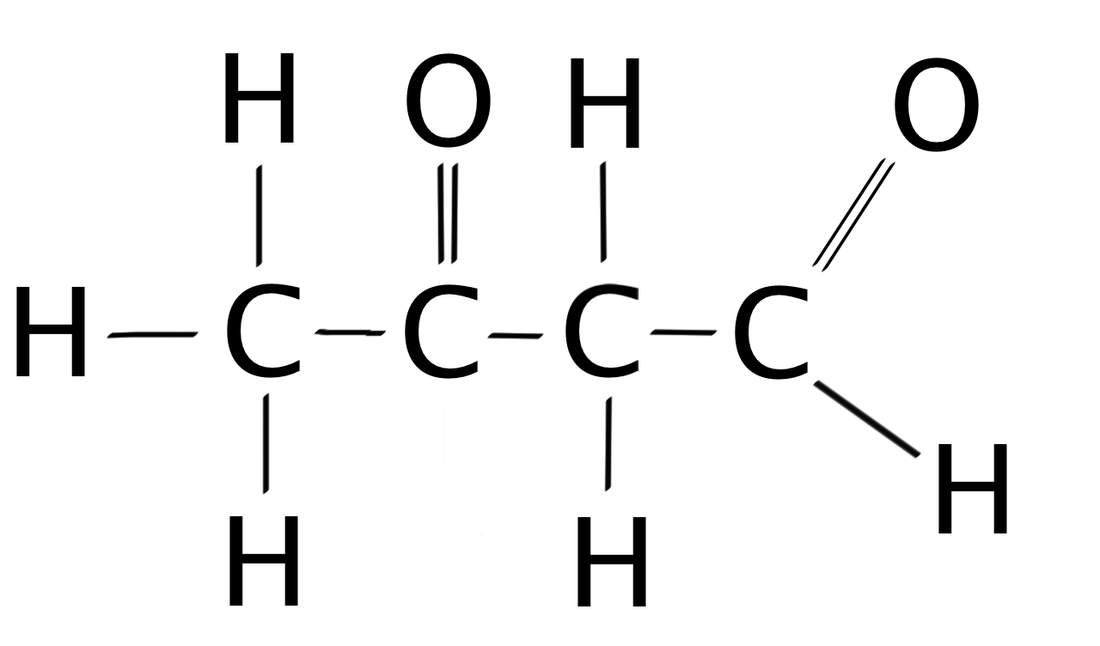
Aldehyde
CnH2n+1CHOe.g. ethanal
CnH2n+1CHOe.g. ethanal
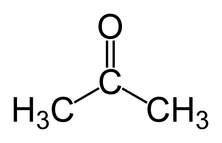
Ketone
CnH2n+1COH e.g propanone or butan-2-one or prefix oxo
Ethanone doesn't exist as it is an aldehyde - ethanal
CnH2n+1COH e.g propanone or butan-2-one or prefix oxo
Ethanone doesn't exist as it is an aldehyde - ethanal
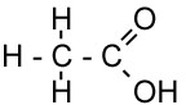
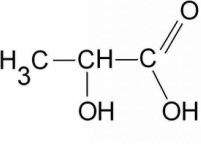
Carboxylic Acid
CnH2n+1COOH e.g. ethanoic acid
CnH2n+1COOH e.g. ethanoic acid
The C=O bond is called the carbonyl group
Ethanol Production
Ethanol is incredibly important because they are common intermediates.
They are species that are easily made and easily convert into other compounds.
It can be made from/via:
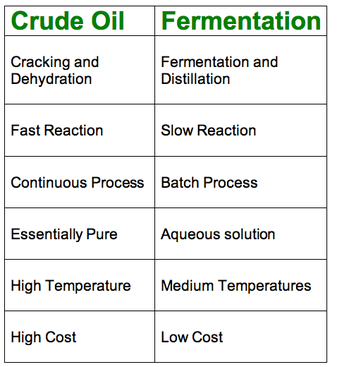
Crude Oil
This is done via Hydration of ethene from crude oil using phosphoric acid and steam in industry
You need to know how sulphuric acid reacts in the mechanism in Alkenes
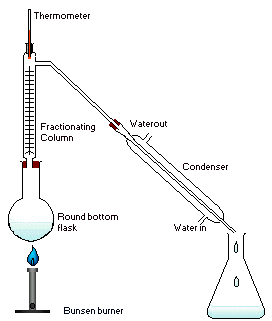
Fermentation
They are species that are easily made and easily convert into other compounds.
It can be made from/via:
Crude Oil
This is done via Hydration of ethene from crude oil using phosphoric acid and steam in industry
You need to know how sulphuric acid reacts in the mechanism in Alkenes
Fermentation
If ethanol is not produced the other possible product is H2O
This needs to occur without access to oxygen so anaerobic respiration can occur
The reaction requires a temperature of 308K (35°C) so the enzymes aren't denatured or ineffective but the reaction can still occur at a fast enough rate
Air is also kept out to prevent oxidation of ethanol to ethanoic acid
Once the solution contains 15% ethanol, the enzymes are unable to function
Ethanol here can be used as a biofuel because it came from renewable crops
This needs to occur without access to oxygen so anaerobic respiration can occur
The reaction requires a temperature of 308K (35°C) so the enzymes aren't denatured or ineffective but the reaction can still occur at a fast enough rate
Air is also kept out to prevent oxidation of ethanol to ethanoic acid
Once the solution contains 15% ethanol, the enzymes are unable to function
Ethanol here can be used as a biofuel because it came from renewable crops
Carbon Neutrality
Carbon neutral is the term given to a process that puts a net value of approximately 0 carbon dioxides into the atmosphere
This happens by the carbon dioxide absorbed at the start is the same as the carbon dioxide produced at the end
Fermentation is a prime example as the carbon dioxide absorbed by the plants is the same amount as the carbon dioxide produced at the end (in theory)
This happens by the carbon dioxide absorbed at the start is the same as the carbon dioxide produced at the end
Fermentation is a prime example as the carbon dioxide absorbed by the plants is the same amount as the carbon dioxide produced at the end (in theory)
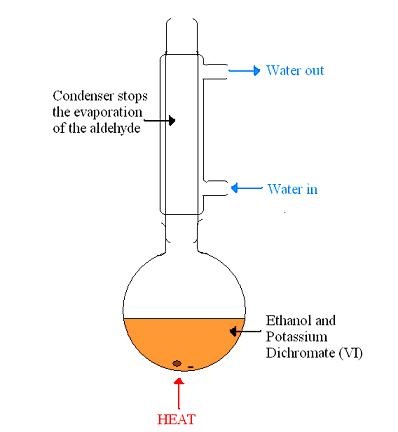
Potassium Dichromate (VI) Reagent
|
Used in the oxidation of alcohols
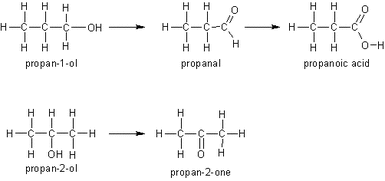
The dichromate (VI) ions are reduced to chromate (III) ions This causes a colour change of orange to green Alcohol - Aldehyde Dilute Sulphuric acid and Potassium Dichromate (VI) is used The mixture is heated and the aldehyde evaporates and distills off the solution The vaporised aldehyde then condenses in a condenser that doesn't create a reflux This only allows partial oxidation CH3CH2OH(l) + [O] → CH3CHO(g) + H2O(l) The [O] is in place of the oxidising agent Alcohol - Aldehyde - Carboxylic Acid Concentrated Sulphuric Acid and excess Potassium Dichromate (VI) is used The mixture is heated, evaporating the aldehyde The vaporised aldehyde then condenses in reflux - the aqueous aldehyde drops back into the solution This is complete oxidation CH3CH2OH(l) + 2[O] → CH3COOH(g) + H2O(l) Alcohol - Ketone This occurs under the same conditions as when an aldehyde is made However it requires a secondary alcohol to do this There is no further oxidation as a ketone cannot be oxidised further |
Tollen's ReagentUsed in the Silver Mirror Test
Tollen's Reagent is a solution of silver nitrate in aqueous ammonia It tests for an aldehyde by oxidising it, creating a carboxylic acid During the test the complex silver (I) ions in the Tollen's reagent are reduced to metallic silver The silver form on the outside of the test tube when the mixture is heated Ketones don't react with the Tollen's reagent |
|
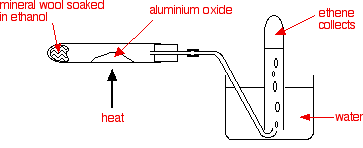
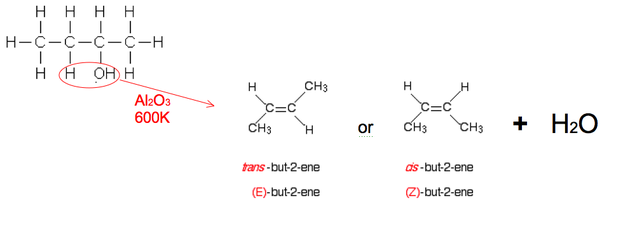
Dehydration
You don't need to know the mechanism for dehydration but you do need to know the effects it has on an alcohol and the different alkenes produced.
It is an elimination reaction when the OH group and an adjacent Hydrogen are removed to form water and an alkene
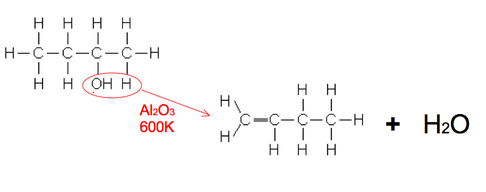
An Aluminium oxide (Al2O3) catalyst is used whilst heated to 600K. (Phosphoric acid (V) is an alternative)
However different alkenes can be formed. Here butan-2-ol forms but-1-ene as well as the two isomers of but-2-ene
Beware! They might continue by asking about one of the products made or about geometric isomerism linking onto other areas of Unit 1. Get an understanding as to where every product can continue onto and its properties.
It is an elimination reaction when the OH group and an adjacent Hydrogen are removed to form water and an alkene
An Aluminium oxide (Al2O3) catalyst is used whilst heated to 600K. (Phosphoric acid (V) is an alternative)
However different alkenes can be formed. Here butan-2-ol forms but-1-ene as well as the two isomers of but-2-ene
Beware! They might continue by asking about one of the products made or about geometric isomerism linking onto other areas of Unit 1. Get an understanding as to where every product can continue onto and its properties.