Energetics
First Law of Thermodynamics - Energy can neither be created or destroyed but can be converted
Standard conditions - 100 kPa, 298K
Standard conditions - 100 kPa, 298K
Enthalpy
Enthalpy (H) - total energy content of a system (higher the enthalpy - more unstable the system is)
It is impossible to measure the total enthalpy but it is possible to measure the change in enthalpy (ΔH) for one mole of that compound
Enthalpy Change - A measure of heat energy given out or taken in during a chemical or physical change at a constant pressure
It is measured in kJ/mol and always requires the + or -
It is impossible to measure the total enthalpy but it is possible to measure the change in enthalpy (ΔH) for one mole of that compound
Enthalpy Change - A measure of heat energy given out or taken in during a chemical or physical change at a constant pressure
It is measured in kJ/mol and always requires the + or -
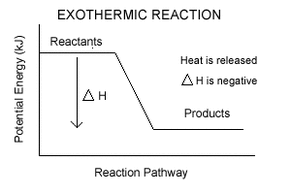
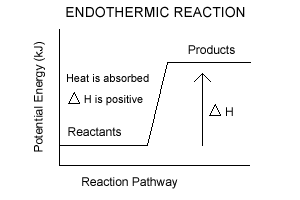
Exothermic Enthalpy ChangeGives out heat
This means the internal enthalpy reduces ΔH is negative Endothermic Enthalpy ChangeTakes in heat
This means the internal enthalpy increases ΔH is positive Don't assume that a reaction is endothermic when heat is put in. Many exothermic reactions need a heat input to start the reaction off |
Enthalpy of Formation
The enthalpy change when one mole of a compound is formed from its constituent elements under standard conditions in its standard states
Given symbol ΔHf°
The product must be one mole at the end
H2 + 1/2O2 → H2O
Given symbol ΔHf°
The product must be one mole at the end
H2 + 1/2O2 → H2O
Enthalpy of Combustion
Bond Dissociation Enthalpy
The enthalpy change when one mole, of a certain bond in a gaseous state, is separated to give independent atoms - averaged over all species containing that bond
Enthalpy of Atomisation
The enthalpy change when all the bonds in one mole of a species are broken under standard conditions in its standard state
Endothermic
Given Symbol ΔHat°
The compound being atomised must be one mole
C2H6 → 2C + 3H2
Endothermic
Given Symbol ΔHat°
The compound being atomised must be one mole
C2H6 → 2C + 3H2
Working out Enthalpy Change
Heat: Measure of energy of all particles
Temperature: Measure of heat energy
Specific Heat Capacity - Energy needed to make 1g of a substance 1K hotter
Energy (J) q = mcΔT (mass heated (g) x specific heat capacity (J/gK) x temp. change (K)
A good way of remembering is by using Cat-muck from cΔTm=q or M-Cat for one side of the equation
To calculate the change in enthalpy use
ΔH = q
mol
Temperature: Measure of heat energy
Specific Heat Capacity - Energy needed to make 1g of a substance 1K hotter
Energy (J) q = mcΔT (mass heated (g) x specific heat capacity (J/gK) x temp. change (K)
A good way of remembering is by using Cat-muck from cΔTm=q or M-Cat for one side of the equation
To calculate the change in enthalpy use
ΔH = q
mol
Displacement Reactions
Working out enthalpy change by using principles from GCSE to principles in amount of substance and enthalpy change
Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s)
To work out the enthalpy change use mcΔT to find out q
You then need to find out which metal is in excess. Use the lower molar value
Then use q
mol
This gives ΔH
Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s)
To work out the enthalpy change use mcΔT to find out q
You then need to find out which metal is in excess. Use the lower molar value
Then use q
mol
This gives ΔH
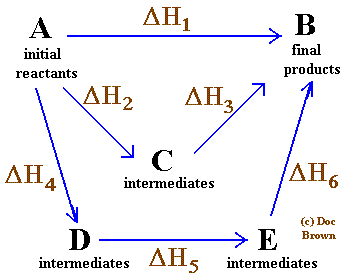
Hess' Law
The enthalpy change for a reaction is the same no matter what route is taken from the reactants to products
ΔH1 = ΔH2 + ΔH3 = ΔH4 + ΔH5 + ΔH6
The cycle used is called a Thermochemical Cycle
The cycle used is called a Thermochemical Cycle
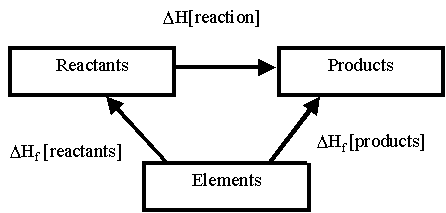
via Enthalpy of Formation
The alternative route is by going to the constituent elements
There is only one problem as to go via the alternative route you must go against the reactants arrow.
This means that you get the equation:
ΣΔHf(products)° - ΣΔHf(reactants)° =ΔH(reaction)°
This means that you get the equation:
ΣΔHf(products)° - ΣΔHf(reactants)° =ΔH(reaction)°
Remember the molar ratio is considered in the enthalpy change value
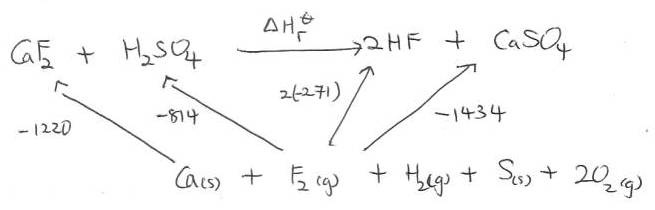
This means ΔH(reaction)° = (2(-271)-1434)-(-1220-814)
= -1976 + 2034
= +58 kJ/mol
= -1976 + 2034
= +58 kJ/mol
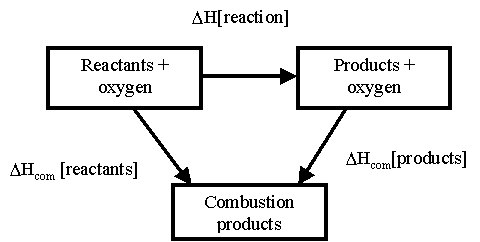
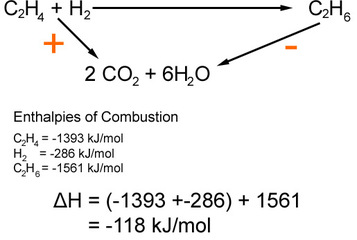
via Enthalpy of Combustion
The reaction takes the alternative route via the combustion products
Again we have a problem as to take this route we have to go against the arrow from the products. This means that to work out the whole enthalpy we use this equation:
ΣΔHc(reactants)° - ΣΔHc(products)° =ΔH(reaction)°
To remember this equation ΔHc = ΔH(reactants) - ΔH(products) use CRaP
with Bond Dissociation Enthalpy
Generally there are too many numbers to work with in a bond dissociation enthalpy thermochemical cycle and so generally just a calculation table is used.
It follows the same equation as combustion
ΣΔHb(reactants)° - ΣΔHb(products)° =ΔH(reaction)°
A good way of not missing bonds is to draw out the displayed formula of each molecule in the reaction
C2H4 + H2 → C2H6
Method
Bond Dissociation Enthalpies
C=C = +611 kJ/mol
C-C = +347 kJ/mol
C-H = +413 kJ/mol
H-H = +436 kJ/mol
Reactants
C=C = 1 x 611
H-H = 1 x 436
C-H = 4 x 412
Total = +2699 kJ/mol
Products
C-C = 1 x 347
C-H = 6 x 412
Total = +2824 kJ/mol
Enthalpy Change
2699 - 2824 = -125 kJ/mol
It follows the same equation as combustion
ΣΔHb(reactants)° - ΣΔHb(products)° =ΔH(reaction)°
A good way of not missing bonds is to draw out the displayed formula of each molecule in the reaction
C2H4 + H2 → C2H6
Method
Bond Dissociation Enthalpies
C=C = +611 kJ/mol
C-C = +347 kJ/mol
C-H = +413 kJ/mol
H-H = +436 kJ/mol
Reactants
C=C = 1 x 611
H-H = 1 x 436
C-H = 4 x 412
Total = +2699 kJ/mol
Products
C-C = 1 x 347
C-H = 6 x 412
Total = +2824 kJ/mol
Enthalpy Change
2699 - 2824 = -125 kJ/mol
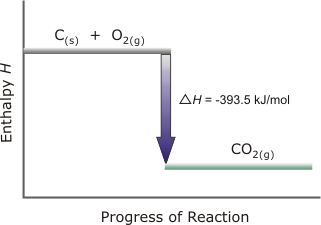
Enthalpy Diagrams
There is an absolute zero which is where the elements sit on an enthalpy diagram
Enthalpy changes then go relative to this
The diagram can also be used the determine the enthalpy change aswell
Below is a very simple enthalpy diagram for the formation of carbon dioxide from carbon and oxygen
Enthalpy changes then go relative to this
The diagram can also be used the determine the enthalpy change aswell
Below is a very simple enthalpy diagram for the formation of carbon dioxide from carbon and oxygen
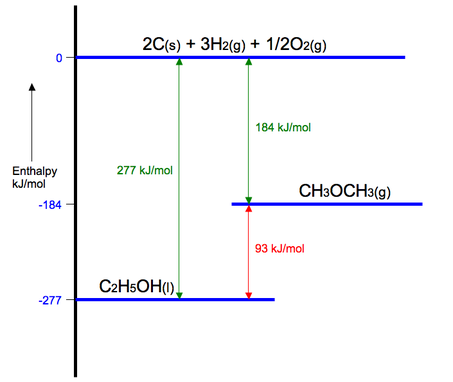
Using the diagram below the enthalpy change of the reaction between methoxymethane to ethanol can be shown to be -93 kJ/mol