Extraction of Metals
Ore is a natural substance containing a large amount of metal
Found as either an oxide, sulphide or carbonate
Found as either an oxide, sulphide or carbonate
Sulphide Ores - Roasting
|
Metal can't be directly removed from a sulphide - they have to be converted into oxides via Roasting
Take Zinc Sulphide: 2ZnO(s) + 3O2(g) → ZnO(s) + 2SO2(g) However a pollutant - sulphur dioxide - is created, which we know from alkanes This problem is solved by turning sulphur dioxide into sulphuric acid which is in high demand |
Oxides are Reduced
|
Not all metal oxide ores are reduced in the same way.
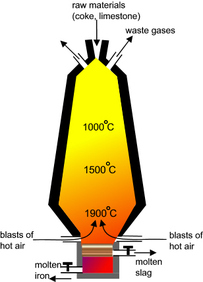
Some are removed by carbon or carbon monoxide which are strong reducing agents in the form of coke (coal) in burnt together in a blast furnace Iron(III) Oxide - HAEMATITE Reduced by carbon or carbon monoxide at 700°C 2Fe2O3 + 3C → 4Fe + 3CO2 Fe2O3 + 3CO → 2Fe + 3CO2 Manganese (IV) Oxide Reduced by carbon or carbon monoxide at 1200°C MnO2 + C →Mn + CO2 MnO2 + 2CO → Mn + 2CO2 |
Carbonates are Reduced
Carbon and Carbon Monoxide are Cheap but not always the best method for extraction
Hydrogen Extracts Tungsten
Electrolysis Extracts Aluminium
|
Aluminium is more reactive than Carbonso can't be reduced by Carbon
Aluminium needs to be molten for electrolysis using graphite electrodes BAUXITE has too high a boiling point of 2050°C It is dissolved in molten CYROLITE to drop the boiling point to 970°C At Cathode Al3+ + 3e- → Al At Anode 2O2- → O2 + 4e- |
Titanium needs different extraction stages
|
Titanium is very abundant but is expensive to produce
Titanium (IV) Oxide - RUTILE Reacted with Carbon and chlorine creating titanium (IV) chloride TiO2 + 2Cl +2C → TiCl4 + 2CO Titanium Chloride is purified via Fraction Distillation in inert atmosphere Argon at 1300K Titanium is reduced at 1273K by a more reactive metal like Sodium or Magnesium TiCl4 + 4Na → Ti + 4NaCl TiCl4 + 2Mg → Ti + 2MgCl2 |
Recycling
Metals can be recycled
This is less expensive than extracting metal from its ore as it requires less energy and no pollutants produced
Scrap Iron can be put to other uses
It can be used to extract copper from a solution
The copper is dissolved in acidified water and is displaced by the Iron
Cu2+ + Fe → Cu + Fe2+
This process gives lower yield than ore reduced by copper
However it is cheaper and better for environment as carbon dioxide isn't produced
This is less expensive than extracting metal from its ore as it requires less energy and no pollutants produced
Scrap Iron can be put to other uses
It can be used to extract copper from a solution
The copper is dissolved in acidified water and is displaced by the Iron
Cu2+ + Fe → Cu + Fe2+
This process gives lower yield than ore reduced by copper
However it is cheaper and better for environment as carbon dioxide isn't produced