Atomic Structure
|
Relative Mass
Relative Charge
|
Proton
1
+1
|
Neutron
1
0
|
Electron
1/1840
-1
|
Protons and Neutrons are held together by a strong nuclear force
Electrons and Protons are held together by electrostatic forces
Electrons and Protons are held together by electrostatic forces
Isotopes
An element with different amount of neutrons
Carbon Dating uses the carbon isotope carbon-14
Carbon-14 is radioactive
This means it has a half life and so organic matter can be dated by the radioactivity levels
Carbon-14 is radioactive
This means it has a half life and so organic matter can be dated by the radioactivity levels
Mass Spectrometer
The abundance of the isotopes can be measured by putting a sample of the element in a Mass Spec
1. Ionisation
|
Electron gun fires electrons through the sample removing one electron from the outer orbital giving an atom +1 charge - this is an ion. +2 ions can also be accidentally formed
Negatively charged plates attract and accelerate the positive ions through a small slit between the plates. This creates a beam
A magnetic field deflects the beam towards the detector. The mass affects the deflection for bigger isotopes require more energy to be deflected
The detector detects the ion as it creates an electric current as it attracts electrons. This creates a mass spectra showing the mass to charge ratio (m/z). This shows the abundance of each isotope. There are also ghost results - this is when a +2 ion is detected and due to its higher charge this means it is much lower than the other detections
|
The RAM is the average of the results
This can be worked out by putting the sum of the abundances over the overall mass used. (When given a percentage assume they are grams in the calculation)
This can be worked out by putting the sum of the abundances over the overall mass used. (When given a percentage assume they are grams in the calculation)
E.g.
90% of 20
10% of 22
(90 x 20) + (10 x 22) = 20.2
100
90% of 20
10% of 22
(90 x 20) + (10 x 22) = 20.2
100
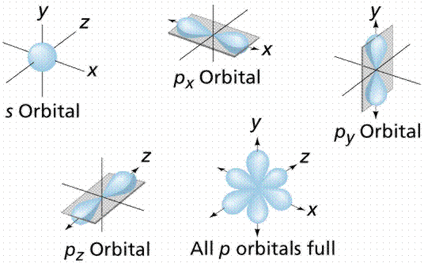
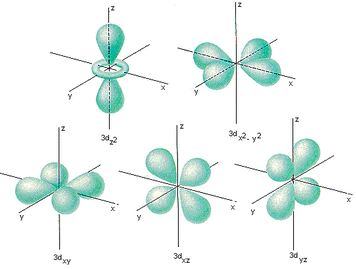
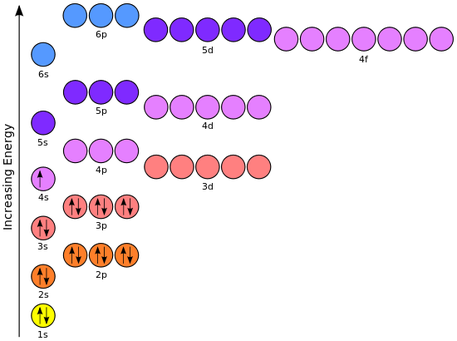
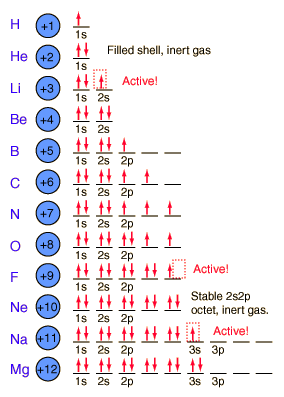
Electron Orbitals
'S' Orbitals - 3D sphere
Nucleus in middle
'P' Orbitals - Dungbells on any axis
Nucleus in between
'D' Orbitals - Two sets of dungbells
Nucleus in between
Electrons move anywhere within the orbital
Only two allowed in each type of orbital
Nucleus in middle
'P' Orbitals - Dungbells on any axis
Nucleus in between
'D' Orbitals - Two sets of dungbells
Nucleus in between
Electrons move anywhere within the orbital
Only two allowed in each type of orbital
Spin
Electrons have a property called 'spin'
Two electrons in the same orbital must have opposite spin
The orbital is most stable when there are two electrons in it
Two electrons in the same orbital must have opposite spin
The orbital is most stable when there are two electrons in it
Ionisation Energy
Ionisation energy is the energy required to remove one mole of electrons from one mole of atoms in a gaseous state
First Ionisation Energy Cu(g) → Cu+ (g) + e-
Second Ionisation Energy Cu+(g) → Cu2+(g) + e-
Second Ionisation Energy Cu+(g) → Cu2+(g) + e-
History of the Atom
1661
1803
b
1896
b
b 1897b
b b 1911
|
Robert BoyleIdea of substances that can't be simplified
John DaltonIdea elements are composed of indivisible atoms
All atoms of the same element have the same mass Atoms of different elements have different masses Henri BecquerelDiscovered radioactivity
Atom is not the fundamental particle JJ ThompsonDiscovered electron
Idea of protons to balance charge Created Plum Pudding Model Ernest RutherfordDiscovered most of the mass is in the centre
Neils BohrDiscovered atom consists of a tiny positive nucleus surrounded by orbiting electrons at different fixed shells around the nucleus
Erwin SchrodingerDiscovered electrons act like waves as well as particles
James ChadwickDiscovered the neutron
|
|
This is not necessarily needed in the exam