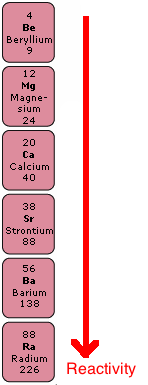
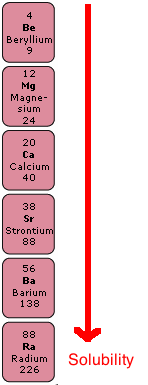
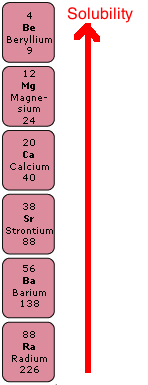
Group 2, the Alkaline Earth Metals
Trends
Atomic Radius
Increase as each time you go down there are extra orbitals. This increases the atomic radius
Boiling Points
Boiling points decrease because the cations get larger. This means the charge density increases relative to the sea of delocalised electrons, which means the metallic bonding is less strong
Ionisation Energies
Ionisation energies decrease going down the group as there are extra orbitals. This increases shielding to the outer electrons and also places them further away. This means the attraction to the nucleus is reduced and so the electron requires less energy to be removed
Chemical Reactions
Reactions with Water
Hydroxides
Sulphates
|
Sulphates are acids
Remember that Sulphate ions have an oxidation state of -2 And Group 2 metal ions have an oxidation state of +2 Barium tests for sulphates
In the test Hydrochloric acid is added to remove carbonate ions. These also ionically bond to Barium forming a white precipitate which affects the observations Barium Chloride solution are added and Barium joins to a sulphate ion. This is a white precipitate |