Equilibria
|
Some reactions are reversible like in the Haber Process.
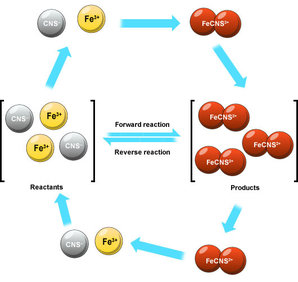
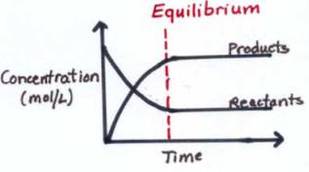
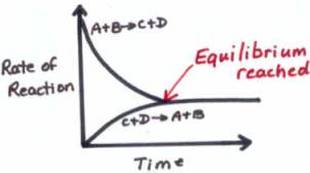
This means the reactants turn into the products whilst the products turn into the reactants. Equilibrium is when the rate of reaction either way don't change. Eventually the forward reactions occur at the same rate as the backwards reaction creating a dynamic equilibrium. This means they are all in even proportion providing an equilibrium mixture. Unlike Kinetics, Equilibria deals with the amount of product The conditions affecting equilbria are like in energetics accept you'll discover that they cause different outcomes |
|
The man with a moustache will now explain this - http://www.youtube.com/watch?v=LBjRDF4XV8Q
|
Chemical Equilibrium
|
A+B ⇌ C + D
If when equilibrium us reached If A + B > C + D then equilibrium lies to the left. If C + D > A + B then equilibrium lies to the right. Equilibrium doesn't have to be half and half During the reaction:
|
Conditions of Equilibrium
- Equilibrium only occurs in a closed system where nothing enters or leaves
- Equilibrium can be approached from either side - doesn't matter whether you start with the products or the reactants
- Is a dynamic process - when the rates of reactions equal each other
Le Chatelier's Principle
Change in . . .
A+B ⇌ C + D
Concentration
Increase A
Forward reaction more likely as there's more reactions occurring on the reactants side
Equilibrium shifts to right
Increase C
Backward reaction more likely
Equilibrium shifts to left
If there is more of something then its rate of reaction increases
Remove C during reaction then the reaction is forced into completion as there's nothing to react in the reverse reaction
Forward reaction more likely as there's more reactions occurring on the reactants side
Equilibrium shifts to right
Increase C
Backward reaction more likely
Equilibrium shifts to left
If there is more of something then its rate of reaction increases
Remove C during reaction then the reaction is forced into completion as there's nothing to react in the reverse reaction
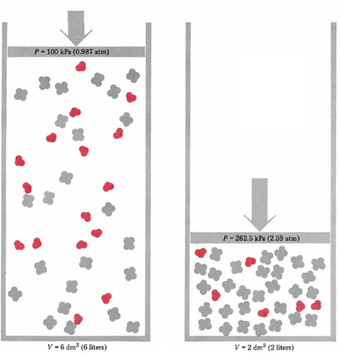
Pressure
|
When the pressure is increased then the equilibrium will shift to the side that has fewer molecules
In the Haber Process the equilibrium shifts to the right as the pressure increases 3H2(g) + 1N2(g) ⇌ 2NH3(g) 4 molecules 2 molecules This is because it means the molecules occupy less space and thus less pressure acts upon them When the number of molecules on either side is the same then equilibrium is not affected 2HI(g) ⇌ H2(g) + I2(g) 2 molecules 2 molecules |
Temperature
If the temperature is increased, equilibrium will move to the endothermic reaction so to get rid of heat
ΔH° -76 kJ/mol →
3H2 + N2 ⇌ 2NH3
← ΔH° +76 kJ/mol
Increase temperature then equilibrium will shift to the left as the backward reaction will increase
ΔH° -76 kJ/mol →
3H2 + N2 ⇌ 2NH3
← ΔH° +76 kJ/mol
Increase temperature then equilibrium will shift to the left as the backward reaction will increase
Catalysts
Catalysts have no affect. They offer an alternative path but don't affect the amount of product