Group 7, The Halogens
Halogen Molecules
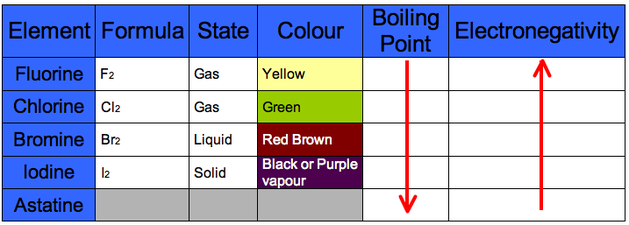
Halogens are Diatomic
Boiling Points increase due to increase in number of electrons meaning an increase in Van der Waal's Forces thus more energy is required to break them
Electronegativity decreases because the attraction to the outer electron decreases due to a larger atomic radius and thus increased shielding. This means the outer electron is easier removed
This also applies to why Iodine is a stronger reducing agent than fluorine and fluorine is a better oxidising agent to iodine
Electronegativity decreases because the attraction to the outer electron decreases due to a larger atomic radius and thus increased shielding. This means the outer electron is easier removed
This also applies to why Iodine is a stronger reducing agent than fluorine and fluorine is a better oxidising agent to iodine
Displacment
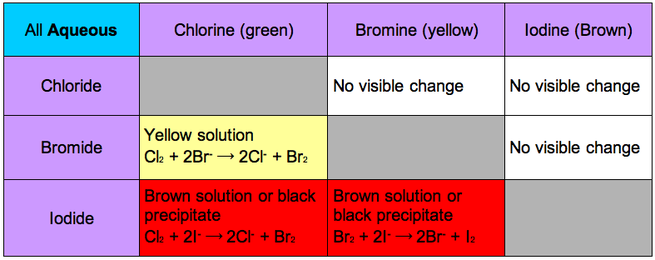
You remember year 8 when you learnt about metals displacing each other. This happens between halogens and halides too
Cl2 + 2NaBr → 2NaCl + Br2
The ionic equation shows chlorine being the oxidising agent
Cl2 + 2Br- → 2Cl- + Br2
Cl2 + 2NaBr → 2NaCl + Br2
The ionic equation shows chlorine being the oxidising agent
Cl2 + 2Br- → 2Cl- + Br2
This is because as you go down the group the attraction to the the outer electron decreases thus the oxidising power decreases as they aren't likely to hold onto the outer electron and are more likely to be oxidised themselves
Chlorine is used to remove Bromide from sea water and Iodide from sea weed
Chlorine is used to remove Bromide from sea water and Iodide from sea weed
Halide Ions Reactions
Beware a Halogen use the prefix -ine e.g. Bromine
However a Halide ion uses the prefix -ide e.g. Bromide
Make sure you use the right one
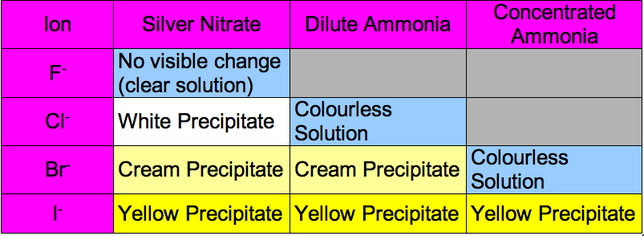
Halide ions generally have a -1 oxidation state
However a Halide ion uses the prefix -ide e.g. Bromide
Make sure you use the right one
Halide ions generally have a -1 oxidation state
In the reaction with silver nitrate a silver halide is formed
In the reaction with ammonia an ammonium halide is formed
We use the ammonia so to tell the difference between the halides as they are not obvious differences
In the reaction with ammonia an ammonium halide is formed
We use the ammonia so to tell the difference between the halides as they are not obvious differences
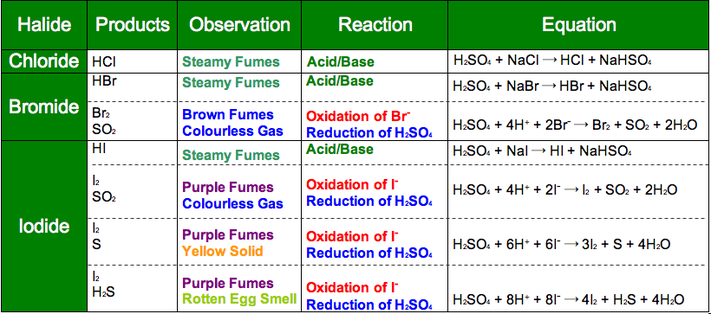
Halide Reactions with Sulphuric Acid
As reducing power increases down the group this trend can be shown via reactions with a salt and sulphuric acid
Two reactions can occur acid/base or redox.
Two reactions can occur acid/base or redox.
Chlorine hasn't got much reducing power so doesn't reduce the Sulphur
Bromine has got some reducing power so reduces Sulphur from +6 to +4
Iodine has got lots of reducing power so reduces Sulphur from +6 to +4 or 0 or -2
Bromine has got some reducing power so reduces Sulphur from +6 to +4
Iodine has got lots of reducing power so reduces Sulphur from +6 to +4 or 0 or -2
Uses of Chlorine
|
Chlorine reacts with Water and Alkalis (aqueous bases)
In Swimming Pools Chlorine reacts with water and is soluble in water It reacts with the below disproportion equilibrium reaction Cl2 + H2O ⇌ HCl + HClO The two products are Hydrochloric acid and Chloric acid. In Hydrochloric acid Chlorine has an OS of -1 In Chloric acid Chlorine has an OS of +1 Chloric acid is a bleach and treats the water by forming chlorate ions which kill bacteria via oxidation HClO + H2O → ClO- + H3O+ The products are acidic so chlorine levels have to be maintained to keep concentration at 0.7 mg/dm^3 However in bright sunlight a different reaction occurs This gives the below reaction Cl2 + H2O → 4HCl + O2 Problems:
With Alkalis Chlorine again disproportionates Cl2 + 2NaOH → NaClO + NaCl + H2O This can then continue onto the below reaction would occur NaClO + H2O → NaOH + HClO This is an alternative to direct chlorination |