Quantum Physics
Photoelectric Effect
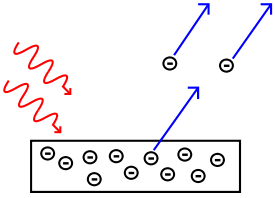
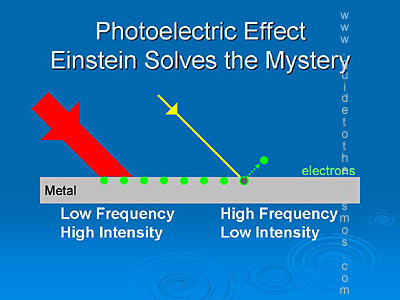
The photoelectric effect is when photons hit a metal passing its energy into the delocalised electrons removing them from the electrostatic forces holding them in the metal. As a photon is a quantum packet of energy as it passes its energy on it no longer exists. Only high frequencies carry enough energy to overcome the work function of the metal - this is called the threshold frequency. However the intensity of the light makes no difference as only one photon can hit an electron so the energy of each electron doesn't change just the amount of electrons being released increases.
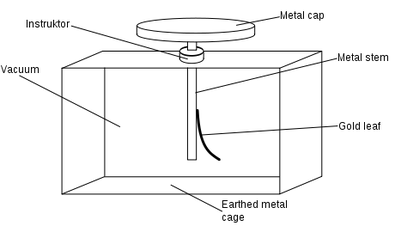
A gold leaf electrosope is used to show the photoelectric effect.
When a negative charge is put through the apparatus then the gold leaf is repelled from the metal bar as they have the same charge.
To get rid of this negative charge UV light can be shone on the metal cap and this removes the excess electrons.
By removing the charge the gold leaf is no longer repelled so returns to the side of the metal bar.
When a negative charge is put through the apparatus then the gold leaf is repelled from the metal bar as they have the same charge.
To get rid of this negative charge UV light can be shone on the metal cap and this removes the excess electrons.
By removing the charge the gold leaf is no longer repelled so returns to the side of the metal bar.
Einstein's Photoelectric Effect Equation
This is why Einstein won the Nobel Prize
Φ is the work function energy. The minimum energy required to release an electron from the metal
Ek is the kinetic energy the electrons have after leaving the metal. This gives how much energy the light is carrying and also enables the frequency to be worked out
Φ is the work function energy. The minimum energy required to release an electron from the metal
Ek is the kinetic energy the electrons have after leaving the metal. This gives how much energy the light is carrying and also enables the frequency to be worked out
E = hf = Ek + Φ
Energy of a photon
A photon is a quantum packet of energy. The energy is measured in eV (electronvolts)
1eV = 1.6 x 10^-19 J
The energy of a photon can be worked out by multiplying the frequency by the Planck's Constant which was created by one of the pioneers in Quantum Physics.
Energy (J) E = hf Planck's Constant (J/s) x frequency (Hz)
Planck's constant is
6.63 x 10^-34 J/s or 4.14 x 10^-15 eV/s
1eV = 1.6 x 10^-19 J
The energy of a photon can be worked out by multiplying the frequency by the Planck's Constant which was created by one of the pioneers in Quantum Physics.
Energy (J) E = hf Planck's Constant (J/s) x frequency (Hz)
Planck's constant is
6.63 x 10^-34 J/s or 4.14 x 10^-15 eV/s
Top Tip: In an exam they might ask you how many photons hit a surface in one second - remember that Hertz means per second - find the frequency
Wave-Particle Duality
De Broglie discovered that all fundamental particles act like a wave as well as a particle
The equation below shows how the wavelength of light is related to the mass of a photon
In an exam the definition is the equation in words
λ = h
mv
Wavelength = Plancks constant
momentum
The masses of the fundamental particles are given in the formula booklet
The equation below shows how the wavelength of light is related to the mass of a photon
In an exam the definition is the equation in words
λ = h
mv
Wavelength = Plancks constant
momentum
The masses of the fundamental particles are given in the formula booklet
Speed of Light
All electromegnetic waves travel at 300'000'000 m/s
The speed is directly proportional to the frequency of the wave and wavelength
v = fλ
This is used for electrons not photons though
For electromagnetic waves this means
c = fλ
c stands for the speed of light
The energy of a photon can also be deduced with the speed and wavelength of it (linking in with wave-particle duality)
E = hc
λ
The speed is directly proportional to the frequency of the wave and wavelength
v = fλ
This is used for electrons not photons though
For electromagnetic waves this means
c = fλ
c stands for the speed of light
The energy of a photon can also be deduced with the speed and wavelength of it (linking in with wave-particle duality)
E = hc
λ
Energy Levels
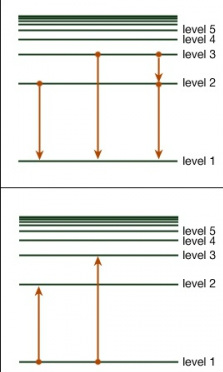
Atoms have energy levels.
These are discrete levels where an electron can sit.
In an exam they'd give you the levels with corresponding energy values. These mean how much energy is required to remove the electron from the atom from that energy level. Ground level always has the lowest highest value.
When an electron moves out of an atom it absorbs energy - this can be absorbed from a photon
When an electron moves down energy levels it releases energy in the form of a photon. The light produced depends on how much energy is given off thus E = hf and E=hc/λ becomes useful
These are discrete levels where an electron can sit.
In an exam they'd give you the levels with corresponding energy values. These mean how much energy is required to remove the electron from the atom from that energy level. Ground level always has the lowest highest value.
When an electron moves out of an atom it absorbs energy - this can be absorbed from a photon
When an electron moves down energy levels it releases energy in the form of a photon. The light produced depends on how much energy is given off thus E = hf and E=hc/λ becomes useful
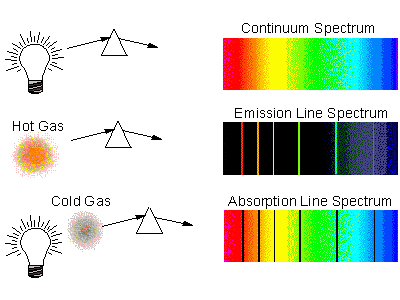
Line Spectra
A line spectra is a display of the wavelengths/frequencies emitted from a light source