Alkanes
Saturated - no double bonds
- General Formula CnH2n+2
- Cycloalkanes CnH2n
- Van der Waal's forces between molecules
- Covalent bonds between atoms
- Not a polar molecule
- As chain increases boiling point rises - due to VDW forces increasing with molecule size
- Insoluble in water
- Low reactivity due to strong bonds
- Combustable
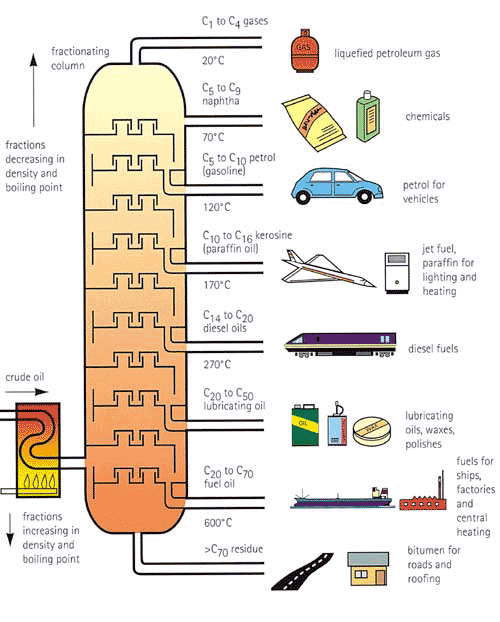
Fractional Distillation
- Crude Oil is a mixture of different Hydrocarbons - from organic material buried in the earth and compressed
- Fractional Distillation separates different length chains (fractions) by their boiling points
- This happens in a Fractionating Column
- The Crude Oil is vaporised
- Fractions rise until they meet a tray just below their boiling point
- This makes that fraction condense whilst smaller fractions continue rising
- At the bottom a thick viscus liquid called bitumen is gathered and at the top is a gaseous methane
You need to know what fractions come at each level and what they are used for
Cracking
Breaks down large chains into smaller alkanes and alkenes which are more useful
There are two ways to crack alkanes
There are two ways to crack alkanes
1. Thermal cracking
- Heated to 700-1200K under 7000kPa
- A pair of electrons in a C-C bond beak to form two hydrocarbon free radicals
- Creates two shorter chains minus one Hydrogen bond (intermediate products)
- One free radical loses a Hydrogen to the other creating an alkane and alkene (you don't need to know this mechanism)
- Alkanes and Alkenes are main products
2. Catalytic Cracking
- Heated to 720K at 1atm
- Zeolite catalyst - honeycomb structure
- Produces Branched alkanes, cycloalkanes and aromatic compounds too
- Catalyst cuts cost and time
Combustion
A fuel is something that can be changed in a reacting vessel to produce useful energy
Fuel + Oxygen → Oxidation Products + Energy Transfer
Complete Combustion
Hydrocarbon + O2 → CO2 + H2O
- Forms CO2 and H2O
- Exothermic reaction
- In balancing:
- Every two Hydrogens in the hydrocarbon gives 1 H2O
- Count up how many oxygen atoms there are and divide it by 2 and place that number in front of the O2 -
because Oxygen is diatomic
Incomplete Combustion
Hydrocarbon + O2 → CO or C + H2O
- Forms CO or just Carbon and Water
- More likely in longer chained hydrocarbons
- Exothermic reaction
- Balanced in same way as Complete Combustion
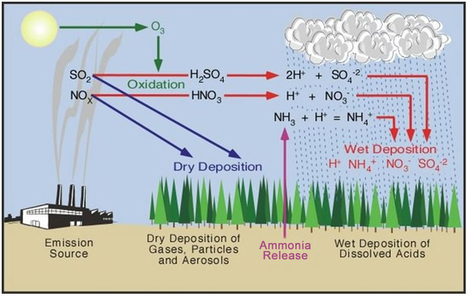
Pollution
- Carbon Monoxide (CO)
- Prevents haemoglobin from bonding with oxygen
- Nitrogen Oxides (NO, NO2, N2O4 or NOx)
- Formed from N2 and O2 when combined under high pressure
- Sulphur Dioxide (SO2)
- Formed from impurities containing sulphur in crude oil
- Carbon Particulates (C)
- Formed by incomplete combustion
- Unburnt Hydrocarbons
- Formed by incomplete combustion
- Carbon Dioxide (CO2)
- Produced through combustion
- Water Vapour (H2O)
- Produced through combustion
Formation of Pollutants
Removal of Pollutants
- Removing Sulphur
- Flue gas desulfurisation
- Catalytic Converters
- Honeycomb platinum and rhodium catalyst
Global Warming
- UV Light penetrates atmosphere warming the ground
- This is re-emitted as infrared radiation
- Greenhouse gases reflect the infrared waves back into the atmosphere warming the ground
- Water is the main greenhouse gas but is a constant level in the atmosphere
- Human implications are raising levels of methane and carbon dioxide through deforestation, burning crude oil etc.