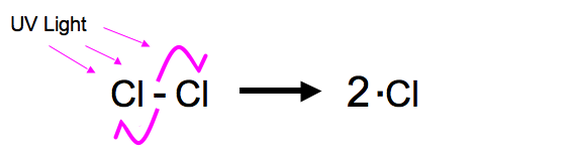Haloalkanes
The general formula is CnH2n+1X where X is a halogen. A halogen is either Fluorine, Chlorine, Iodine and Bromine with their appropriate prefixes when named. Haloalkanes are polar molecules with the halogen being δ- (delta negative).
In mechanisms the curly arrows represent the movement of a free pair of electrons.
In mechanisms the curly arrows represent the movement of a free pair of electrons.
Nucleophilic Substitution
A nucleophile is a reagent that attack and form bonds with a polar molecule. In Halogens the nucleophile replaces a halogen in a haloalkane. Nucleophiles have a spare pair of electrons. So you have:
A good point to take note of is that tertiary haloalkanes are more likely to take part in nucleophilic substitution. The mechanisms and overall formulas are in the file below:
- :OH-, Hydroxide ion in an aqueous state, the product is an alcohol
- :NH3, Ammonia, the product is an amine
- :CN-, Cyanide ion, the product is a nitrile
A good point to take note of is that tertiary haloalkanes are more likely to take part in nucleophilic substitution. The mechanisms and overall formulas are in the file below:
| Haloalkanes Mechanisms | |
| File Size: | 369 kb |
| File Type: | rtf |
Elimination Reaction
In this reaction potassium hydroxide (alcoholic) reacts with a haloalkane to produce an alkene, water and a potassium reacted with the halogen. This is where it differs from the aqueous potassium hydroxide. In this reaction KOH acts as a base. Tertiary haloalkanes tend towards elimination.
The mechanism and overall formula for this type of reaction is in the file below:
The mechanism and overall formula for this type of reaction is in the file below:
| Elimination Mechanism | |
| File Size: | 48 kb |
| File Type: | doc |
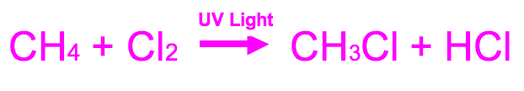
Free Radical Substitution
A free radical is an atom either by itself or part of a molecule that has a free electron ready to form a covalent bond.
They are very reactive and attack other molecules so to complete a covalent bond with the free radical.
The overall formula is:
They are very reactive and attack other molecules so to complete a covalent bond with the free radical.
The overall formula is:
However, there are THREE steps in free radical substitution. They are initiation, propagation and termination.
1. Initiation
In this reaction a halogen is broken from its diatomic state by UV light to create two halogen free radicals. If we take Chlorine (which is most common) it will create two chlorine free radicals. This process is called Homolytic Fission. This mechanism is shown below
2. Propagation
The whole point in this reaction is to get a haloalkane. There are two reactions in the propagation stage. the first involves one chlorine free radical bonds with an alkane to form hydrochloric acid and free radical organic compound. This then reacts with a chlorine molecule (Cl2) forming a haloalkane and another chlorine free radical. The latter can then take part in the first reaction creating a cycle. You don't need to know the mechanism for these reactions.
1) CH4 + ·Cl → HCl + ·CH3
2) ·CH3 + Cl2 → CH3Cl + ·Cl This final free radical joins in the first part of the propagation step.
1) CH4 + ·Cl → HCl + ·CH3
2) ·CH3 + Cl2 → CH3Cl + ·Cl This final free radical joins in the first part of the propagation step.
3. Termination
There is one problem with having all these highly reactive free radicals is that they will react with anything so they won't always react with a the intended molecule in the propagation stage. The termination reactions mean that there are no more chlorine radicals left as products to carry on reacting
1) ·Cl + ·Cl → Cl2
2) ·CH3 + ·CH3 → C2H6
3) ·CH3 + ·Cl → CH3Cl
By increasing the amount of methane molecules there is less chance of a free radical not bonding with one to start off propagation thus decreasing the chance of the above reactions occurring.
1) ·Cl + ·Cl → Cl2
2) ·CH3 + ·CH3 → C2H6
3) ·CH3 + ·Cl → CH3Cl
By increasing the amount of methane molecules there is less chance of a free radical not bonding with one to start off propagation thus decreasing the chance of the above reactions occurring.