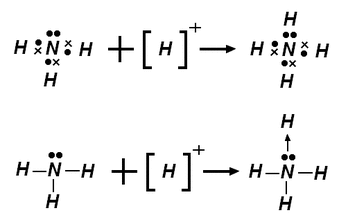Bonding
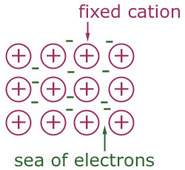
Metallic Bonding
- Delocalised electrons are free to move and carry charge through the structure creating a current
- Electrostatic forces hold the structure together
- This means metallically bonded compounds have high boiling points
- Uniform structure allows for malleability and ductility because ions slide past each other as they are the same size
- Alloys affect properties because the ions are different sizes
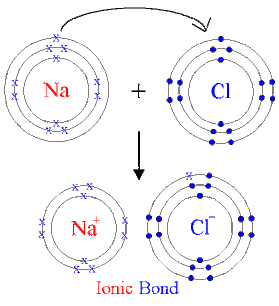
Ionic Bonding
- Between metals and non-metals
- Metal donates electron to create a full outer orbital on the non-metal
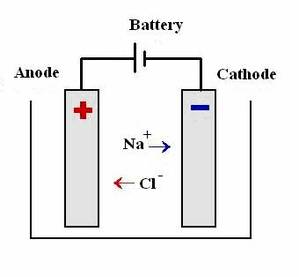
- Takes part in electrolysis
- Creates a current when connected to a power source in an aqueous state
- The opposite forces mean it has a high boiling point
- It is a brittle compound because when bonds are moved they repel each other so compounds break up
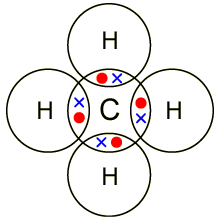
Covalent Bonding
- Electrons are shared as a pair
- When one pair of electrons are shared this forms a sigma bond
- Sometimes electron deficient molecules attach themselves to electron rich molecules forming a dative or coordinate covalent bond. This is when both electrons in the bond come from one molecule. When drawn an arrow is pointing towards the electron deficient molecule to show that both electrons have come from one molecule
Electronegativity
The ability of an element to attract electron density towards itself in a covalent bond
Electronegativity increases across a period and up the groups
Helium and noble gases are not electronegative as they don't form covalent bonds
Electronegative elements are labelled as δ-
Helium and noble gases are not electronegative as they don't form covalent bonds
Electronegative elements are labelled as δ-
Electronegative elements:
Nitrogen
Oxygen
Fluorine
Chlorine
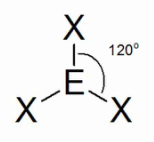
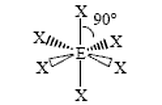
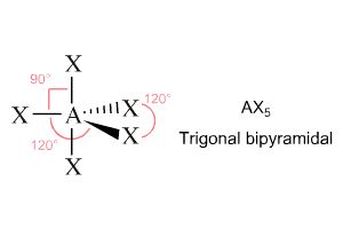
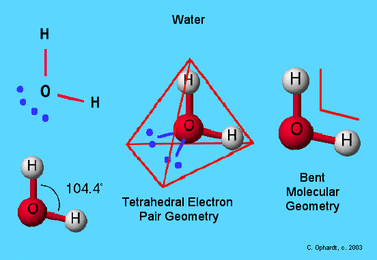
Shapes
A straight line means the bond is on the same plane
A wedge means it is coming towards you
A dashed line means it's going away
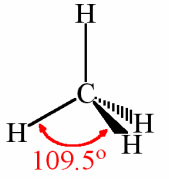
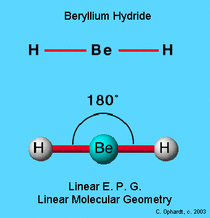
Need to know structure name and bond angle
A wedge means it is coming towards you
A dashed line means it's going away
Need to know structure name and bond angle
Tetrahedral
Linear
Trigonal Planar
Octahedral
Trigonal Bipyramidal
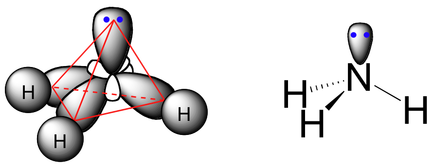
Lone Pair Repulsion
The lone pair of electrons repel the other bonding pairs because they are closer to the central atom thus repelling the other bonds more.
The general rule of thumb is each lone pair reduce the bond angle by 1.5º
Only atoms are taken into account when naming the shape
The general rule of thumb is each lone pair reduce the bond angle by 1.5º
Only atoms are taken into account when naming the shape
Triangular Pyramid
Bonding angle is 107º
V Shaped / Bent
Intermolecular Forces
Intermolecular means between molecules
Intramolecular means within molecules
Intramolecular means within molecules
- Van der Waal's Forces
- When atoms are attracted to each other because of opposite charges
- This occurs because electrons in an atom are unlikely to be evenly spread - this is called a temporary dipole
- When approaching another atom it will cause a temporary dipole in that atom because equal charges repel. This is an induced dipole
- These are weak forces
|
Temporary Dipole
|
Second atom has an induced dipole
|
Factors
This can be shown by looking at the standard states of group 7 elements
Fluorine - Gas
Chlorine - Gas
Bromine - Liquid
Iodine - Solid
This shows that with the increase of electrons and size of atoms the boiling points increase because its harder to break the bonds of the larger elements
- The number of electrons
- The mass/size of the atom
- How close they can get together - branched molecules impede attraction and so have weaker VDW forces
This can be shown by looking at the standard states of group 7 elements
Fluorine - Gas
Chlorine - Gas
Bromine - Liquid
Iodine - Solid
This shows that with the increase of electrons and size of atoms the boiling points increase because its harder to break the bonds of the larger elements
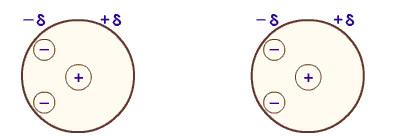
- Dipole-Dipole Forces
- Occurs when there is a permanent dipole - when certain atoms are δ negative
- This means the atoms are permanently electronegative
- Electronegative elements are Nitrogen, Oxygen, Fluorine and Chlorine
- Larger than VDW forces
Factors
- Requires electronegative atoms to create a charge difference
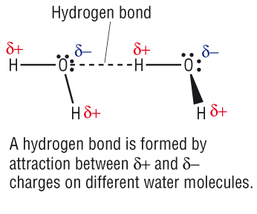
- Hydrogen Bonding
- When a free pair of electrons are attracted towards a δ positive Hydrogen
- Only occurs between Hydrogen and either Nitrogen, Oxygen or Fluorine
- Only occurs between covalently bonded molecules
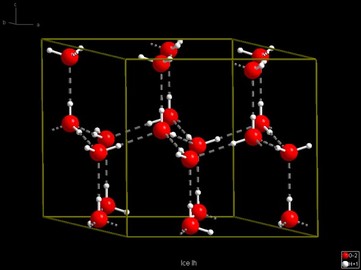
Ices density is less than water's because the H bonds increase in length so to keep the water's bond angles the same.
This means Hydrogen Bonded molecules always dissolve in water
Hydrogen Bonds are also in DNA
Hydrogen Bonds are also in DNA
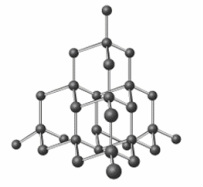
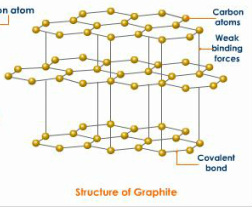
Carbon Allotropes
Allotropes are different forms of carbon only structures
- Diamond
- Graphite
|