Kinetics
The study of factors that affect the rate of a chemical reaction
Everything links in with itself in this chapter - read on if something is mentioned and not explained, it is later on
Everything links in with itself in this chapter - read on if something is mentioned and not explained, it is later on
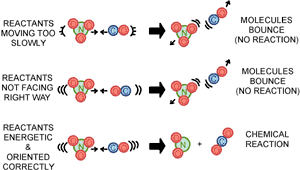
Collision Theory
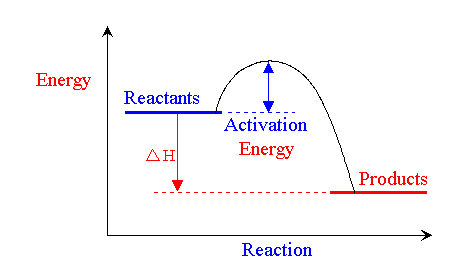
Activation Energy
The minimum energy required for a chemical reaction to occur (the energy required to break the bonds)
Given symbol Ea
The point a reaction occurs is called the transition point or activation complex
Given symbol Ea
The point a reaction occurs is called the transition point or activation complex
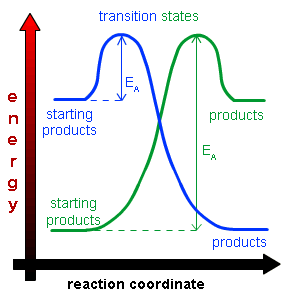
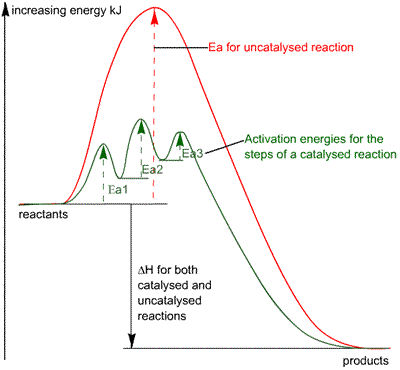
Exothermic and Endothermic Activation energy
Both require activation energy but the products and reactants start have different placed enthalpies
Both require activation energy but the products and reactants start have different placed enthalpies
Factors in Chemical Reactions
Temperature
Increase in temperature means the particles vibrate more, move round more meaning a greater chance of successful collisions
Concentration and Pressure
Increase in concentration means the particles are closer together meaning a greater chance of successful collisions
Surface Area
Increase in surface area means more particles available to collide and so there's a greater chance of successful collisions
Catalyst
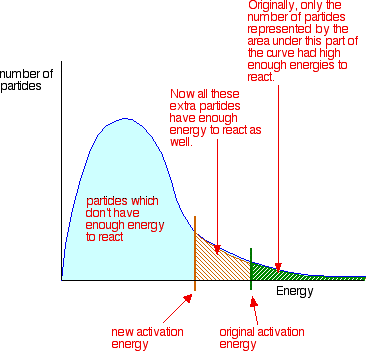
Lowers the activation energy for a reaction and so more particles are available to react - refer to maxwell-boltzmann distribution - and so increases the chance of a successful collision
Increase in temperature means the particles vibrate more, move round more meaning a greater chance of successful collisions
Concentration and Pressure
Increase in concentration means the particles are closer together meaning a greater chance of successful collisions
Surface Area
Increase in surface area means more particles available to collide and so there's a greater chance of successful collisions
Catalyst
Lowers the activation energy for a reaction and so more particles are available to react - refer to maxwell-boltzmann distribution - and so increases the chance of a successful collision
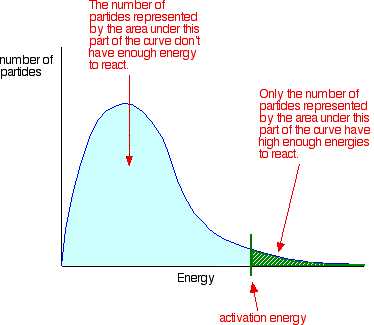
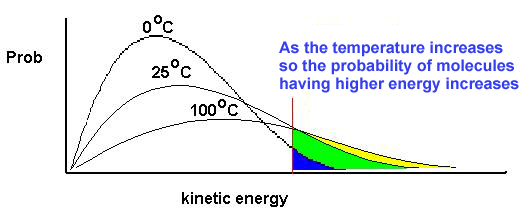
The Maxwell-Boltzmann Distribution
Not all particles have the same amount of energy. The Maxwell-Boltzmann distribution shows that the majority of particles don't have enough energy to overcome the Activation Energy. This shows the reason for needing to change the factors
Temperature's affect on the MBD
Catalysts affect on the MBD
Catalysts
Catalysts do take part in a reaction but they are chemically unchanged in the overall reaction
Heterogenous Catalyst - catalyst is in a different state or phase to reactants
Homogenous Catalyst - catalyst is in same state or phase to reactants
Types of Catalyst
Haber Process - Iron (II) - heterogenous - making fertilisers go to equilibria
Making Nitric Acid via Ostwald Process - Platinum and Rhodium - heterogenous - fertilisers and explosives
Hardening fats - Nickel - heterogenous - making margarine go to alkenes
Carbon cracking - Zeolite - heterogenous - petrol go to alkanes
Catalytic Converter - Platinum and Rhodium - heterogenous - removing pollutants go to alkanes
Hydration of ethene - Phosphoric acid (Industry)/Sulphuric Acid - heterogenous - making ethanol go to alkenes and alcohols
Esterification - Hydrogen ion - homogenous - making solvents
Making Nitric Acid via Ostwald Process - Platinum and Rhodium - heterogenous - fertilisers and explosives
Hardening fats - Nickel - heterogenous - making margarine go to alkenes
Carbon cracking - Zeolite - heterogenous - petrol go to alkanes
Catalytic Converter - Platinum and Rhodium - heterogenous - removing pollutants go to alkanes
Hydration of ethene - Phosphoric acid (Industry)/Sulphuric Acid - heterogenous - making ethanol go to alkenes and alcohols
Esterification - Hydrogen ion - homogenous - making solvents
Catalytic Converters
The converter is a platinum and rhodium honeycomb catalyst. They enable harmful products to to react with each other and create less harmful gases
carbon monoxide + nitrogen oxides → nitrogen + carbon dioxide
hydrocarbons + nitrogen oxides → nitrogen + carbon dioxide + water
Adsorption - The gases form weak bonds with the catalyst holding them in the right position to react
Desorption - Gases then break away
carbon monoxide + nitrogen oxides → nitrogen + carbon dioxide
hydrocarbons + nitrogen oxides → nitrogen + carbon dioxide + water
Adsorption - The gases form weak bonds with the catalyst holding them in the right position to react
Desorption - Gases then break away