Introduction to Organic Chemistry
Functional Group - Tells the homologous series (family) the molecule belongs to
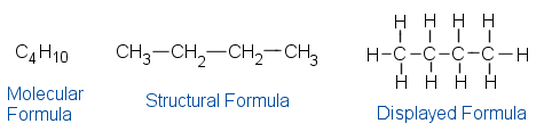
Displayed Formula - Relative positioning of all the atoms in a molecule and the bonds between them
Displayed Formula - Relative positioning of all the atoms in a molecule and the bonds between them
The best way to crack nomenclature (naming) and isomerism is through practice and doing examples
Nomenclature
Prefixes for naming organic compounds go as below
|
Prefix
meth eth prop but pent hex hept oct non dec |
No. Carbons in the longest chain
1 2 3 4 5 6 7 8 9 10 |
Suffixes for organic compounds going by the homologous series
|
Suffix
ane ene anol yl |
Homologous Series
alkane C-C alkene C=C alcohol C-O-H alkyl (branched alkane) |
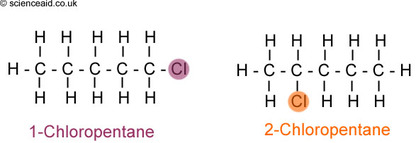
Naming Haloalkanes
Prefixes
Cl - Chloro
Br - Bromo
I - Iodo
Cl - Chloro
Br - Bromo
I - Iodo
Naming Process
- Find Longest Chain
- Identify functional groups added and on which carbon
- (2) is the prefix, (1) is the suffix
- If more chains
- If more than one of the same group then label what number carbons they are on and put in the prefix on the prefix di for 2
of the same functional groups, tri for 3 and tetra for 4 (ignore these in the alphabetical order)
Isomerism
- Positional Isomerism
Functional groups in different positions but have the same molecular formula but different structural and displayed formula
- Chain Isomerism
Same molecular formula but different name as there is a change in the longest chain length
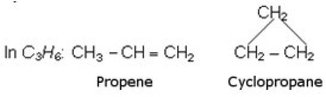
- Functional Isomerism
Same molecular formula but different functional group
In the exam this generally arrives as an alkene functional isomer which becomes a cycloalkane