Alkenes
Alkenes are an organic molecule with the general formula CnH2n. At first glance the difference between alkanes and alkenes isn't very big but in fact that double bond makes an astronomically big difference. The difference is in the shape; remember them all the way back in Unit 1?
Structure
|
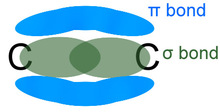
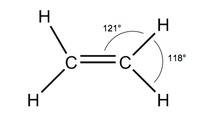
In alkanes there was only C-C bonds but now this has changed as there is now a C=C bond involved. The reason alkanes were nice was because every bond had free rotation so it didn't matter where you put anything around each carbon molecule because it would still be the same. However, in alkenes the double bond prevents this. This is because of the difference in orbitals around each bond.
In alkanes there is only sigma bonds which allow for free rotation but because of the extra bond between the two carbons a new type of bond is formed called a pi bond (remember that number used in circles for Maths?) instead of two sigma bonds. This restricts free rotation which means there is now a difference as to where different atoms are placed. This can be shown more clearly when naming haloalkenes. |
NO FREE ROTATION
The double bond pushes the other two bonds further away and closer to each other
|
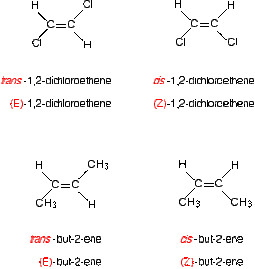
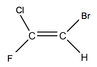
Geometric Isomerism
Because there is no free rotation it matters as to what side carbon chains and halogens join. Because of this when naming this causes problems. There is either the prefix Z or E. A Z isomer is when the two groups are on the Zame side whereas an E isomer is when they aren't on the same side. This affects the molecules shape as the which side they are. If however one side of the double bond has two functional groups the same then there is no isomerisation.
|
To the left shows geometric isomerisation.
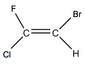
The the right shows molecules that aren't isomers because they have the same functional groups However when the elements on one carbon are different then the highest mass element is what is taken account of. E.g. Chlorine is bigger than Fluorine so would be taken account of during the naming
When there is an OH group in an alkene it is referred to with the prefix Hydroxyl |
Electrophilic Addition
In electrophilic addition, electrophiles are attracted towards alkenes. They cause the bond to break creating a carbocation which is a positive ion as a carbon is missing one sigma bond. This then bonds with the remaining halogen left behind which was originally bonded in the electrophile in the first place.
Hydrolysis / Electrophilic Addition
(In industry a H3PO4 catalyst in steam at 413K or 140°)
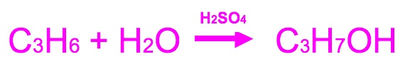
H2SO4 heated is the mechanism used
The overall reaction however is different to the mechanism.
The overall reaction is:
H2SO4 heated is the mechanism used
The overall reaction however is different to the mechanism.
The overall reaction is:
In the overall reaction concentrated sulphuric acid acts as the catalyst with propene reacting with water to create propanol.
The Mechanisms for both electrophilic addition reactions are below:
The Mechanisms for both electrophilic addition reactions are below:
| Electrophilic Mechanisms | |
| File Size: | 385 kb |
| File Type: | rtf |
To reverse this reaction the process of dehydration is used using Al2O3 and heat
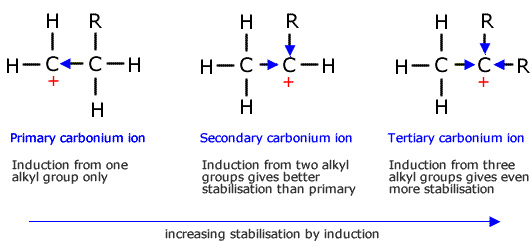
Carbocations
Carbocations are hydrocarbons that have a positive charge on one of the carbons. They are an ion and an intermediate molecule
They can either be primary, secondary or tertiary. When formed they are more likely to for a tertiary carbocation for it is more stable.
Tertiary carbocations are more stable due to the inductive effect.
They can either be primary, secondary or tertiary. When formed they are more likely to for a tertiary carbocation for it is more stable.
Tertiary carbocations are more stable due to the inductive effect.
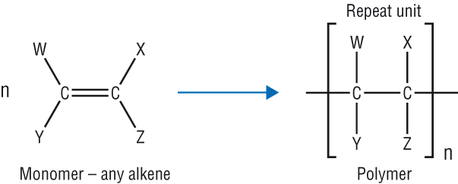
Polymerisation
Alkenes polymerise as they join onto each other in endless chains.
Adding alkenes to each other is called addition polymerisation (not additional)
The original alkene is called the monomer
The smallest unit that is repeated is called the repeating unit
In nomenclature they are named by putting poly in front of the monomer. e.g. poly(propene)
The fact the double bond is removed means that polymers aren't reactive which means they aren't biodegradable
Adding alkenes to each other is called addition polymerisation (not additional)
The original alkene is called the monomer
The smallest unit that is repeated is called the repeating unit
In nomenclature they are named by putting poly in front of the monomer. e.g. poly(propene)
The fact the double bond is removed means that polymers aren't reactive which means they aren't biodegradable